Promus ELITE™
Everolimus-Eluting Platinum Chromium Coronary Stent System
PLATINUM Workhorse Trial
Study Objective
Evaluate the safety and effectiveness of the Promus PtCr EES Coronary Stent System* for the treatment of patients with up to 2 de novo lesions ≤ 24 mm in length; ≥ 2.50 mm to ≤ 4.25 mm in diameter compared to the Xience CoCr EES.
Study Design
Prospective, Randomized, Controlled, Non-inferiority, Multicenter
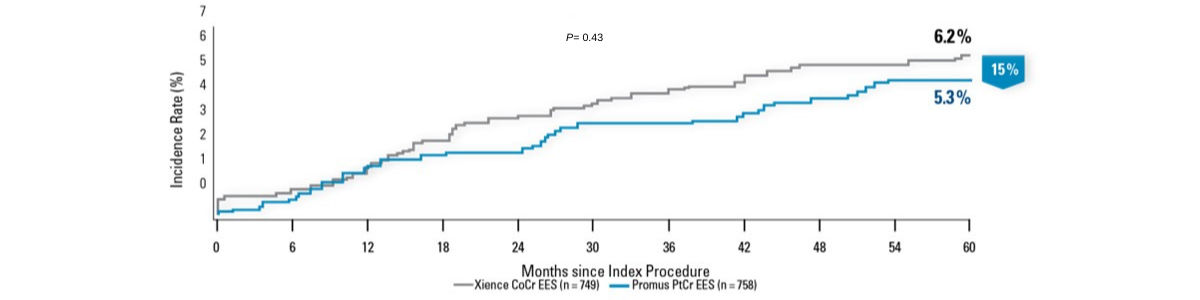
Numerically Lower Ischemia-Driven TLR Through 5 Years2
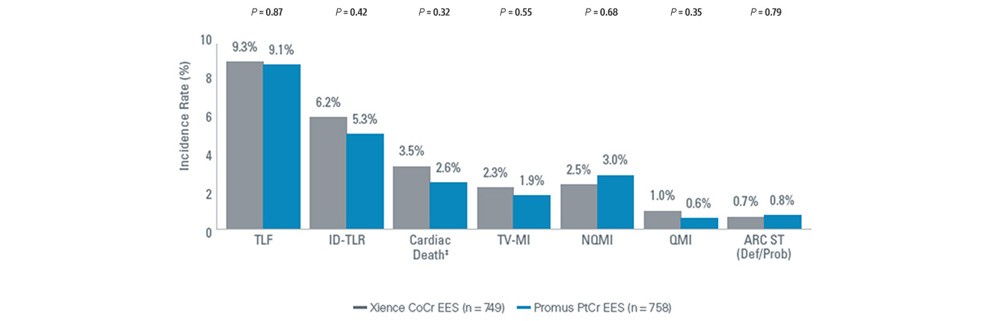
Comparable Event Rates Through 5 Years2