Clinical Studies

WATCHMAN™ LAAC Device: New PREVAIL 5 Year Data from TCT and 5 Year Meta-Analysis results published in JACC

TCT 2017:
FAME 2 Trial 3-year Outcome and
cost-effectiveness of FFR

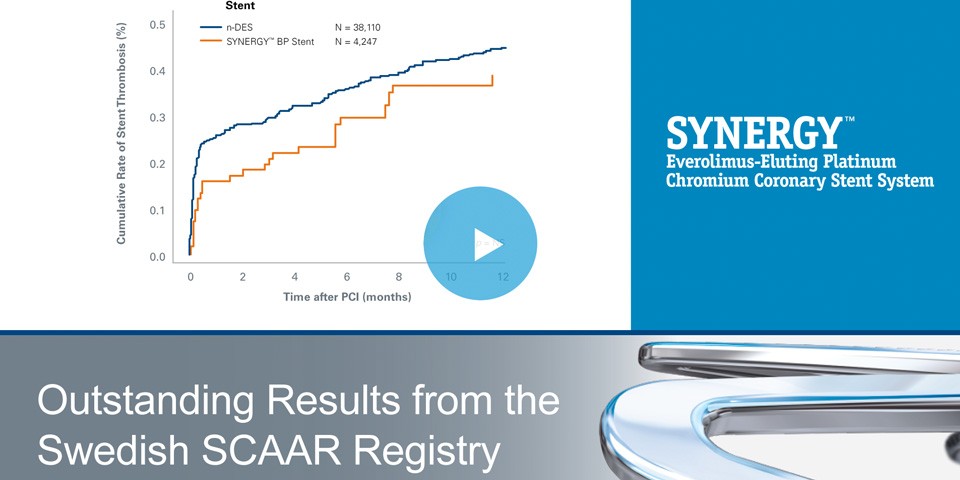
SYNERGY™ BP Stent: outstanding results from the Swedish SCAAR Registry
* Two strokes in PREVAIL are excluded because the baseline MRS score was unavailable
1. Reddy VY, 5-Year Outcomes After Left Atrial Appendage Closure From the PREVAIL and PROTECT AF Trials. DOI: 10.1016/j.jacc.2017.10.021
2. Sarno, G., et al. Cathet. Cardiovasc. Intervent.. doi:10.1002/ccd.27030














