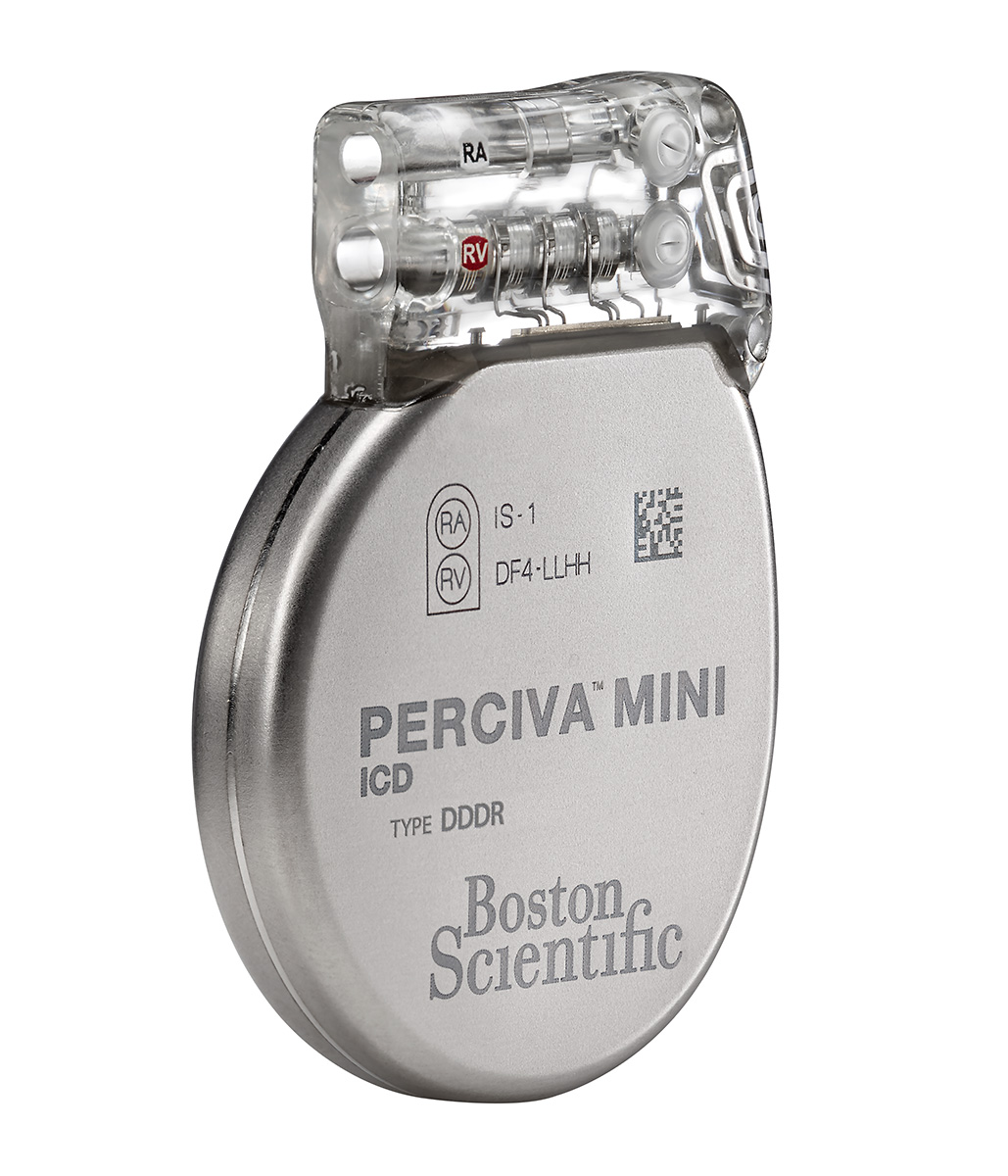
PERCIVA™ ICD
Models D400, D401, D412, D413
Introducing the PERCIVA™ family of ICDs offering an extraordinary, holistic approach to cardiac care.
Key Resources

Product Details
Models D400, D401, D412, and D413
- Projected to last up to 9.3 years for VR devices and 8.5 years for DR devices.*
- This small (28.5 cc) and thin (9.9 mm) high-energy device is designed to enhance patient comfort.
- IMAGEREADY™ MR-Conditional Systems allow patients to receive full body MR-Conditional scans at 1.5T or 3T without exclusion zone, scan duration, or patient height restrictions.**
- Atrial Arrhythmia Report provides a comprehensive view of your patient’s AT/AF status and information for assessing AFtreatment efficacy.
- RightRate™ features Boston Scientific’s MV Sensor — the only sensor clinically proven to restore chronotropic competence.1
- The PaceSafe™ RV, RA, and LV auto threshold feature is designed to maintain adequate output safety margins.
- Offers AcuShock™ Advanced Technology, multiple programmable options to reduce inappropriate and unnecessary shocks,including a choice of rhythm discriminators, RhythmID™ with RhythmMatch™ (customization of Rhythm ID algorithm),antitachycardia pacing (ATP) therapy in all rates zones, and advanced sensing and filtering.
- Wireless ECG saves time and simplifies follow-up.
- EasyView™ header and color coded lead ports designed to make the implant experience more efficient.
- SafetyCore™ technology is intended to provide lifesaving shock therapy and basic pacing functionality in the event of an unrecoverable fault.
* Assumes: 2.0V RA, 2.0V RV, 0% pacing, 700Ω, No LATITUDE, No Respiratory Rate Sensor, No Heart Failure Sensor Suite.
** When conditions of use are met.
Mechanical Specifications
| Model | Type | Size (cm) (W x H x D) | Mass (g) | Volume (cc) | Connector Type | MR Conditional | C-Codes |
|---|---|---|---|---|---|---|---|
| D400 | VR | 5.23 x 7.14 x 0.99 | 61.9 | 28.5 | RV: IS-1/DF-1 | No | C1722 |
| D401 | DR | 5.23 x 7.14 x 0.99 | 62.3 | 28.5 | RA: IS-1; RV: IS-1/DF–1 | No | C1721 |
| D412 | VR | 5.23 x 6.71 x 0.99 | 60.0 | 26.5 | RV: DF4 | Yes | C1722 |
| D413 | DR | 5.23 x 7.03 x 0.99 | 62.5 | 28.0 | RA: IS-1; RV: DF4 | Yes | C1721 |
Longevity Information
Projected Longevitya |
Type | Pacing Amplitude | Pacing | 500Ω with LATITUDE™b |
700Ω with LATITUDE™b | 900Ω with LATITUDE™b | 700Ω no LATITUDE™, RS, or HFSSc |
|---|---|---|---|---|---|---|---|
| VR | |||||||
| Typical programmed setting | VR | 2.5 V | 15% | 8.0 | 8.0 | 8.1 | 9.1 |
| Maximum labeled longevity | VR | 2.0 V / Off | 0% | 8.2 | 8.2 | 8.2 | 9.3 |
| DR | |||||||
| Typical programmed setting | DR | 2.5 V | 15% | 7.2 | 7.3 | 7.3 | 8.1 |
| Maximum labeled longevity | DR | 2.0 V | 0% | 7.5 | 7.5 | 7.6 | 8.5 |
- Assumes 70 PPM LRL; DDDR mode; 0.4 ms Pulse Width (RA, RV); sensors On, Heart Failure Sensor Suite On.
- Projected longevity is calculated assuming 2 maximum energy charging cycles per year, including automatic capacitor re-forms and therapeutic shocks. These calculations also assume 3-channel EGM Onset is on and that the pulse generator spends 3 months in Storage mode during shipping and storage.
a. Assumes ZIP telemetry use for 2 hours at implant and for 40 minutes annually for in-clinic follow-up checks.
b. Assumes standard use of the LATITUDE™ Communicator as follows: Daily Device Check on, quarterly scheduled remote follow ups, and other typical interrogations.
c. Assumes LATITUDE™ Communicator is not used, Respiratory Sensor is Off, and Heart Failure Sensor Suite is Off.
Additional Longevity Information
- Warranty information available at www.bostonscientific.com/warranty
- Devices use Li/MnO2 chemistry.
- The Usable Battery Capacity is 1.0 Amp-hours (typical implant to battery capacity depleted).
- Shelf life is 2 years (before use by).