IMPERIAL Clinical Trial Results
The IMPERIAL RCT demonstrated that Eluvia DES is clinically effective and safe in treating patients with symptomatic SFA disease, both in the short-term during the height of restenosis risk, and long-term out to five years.
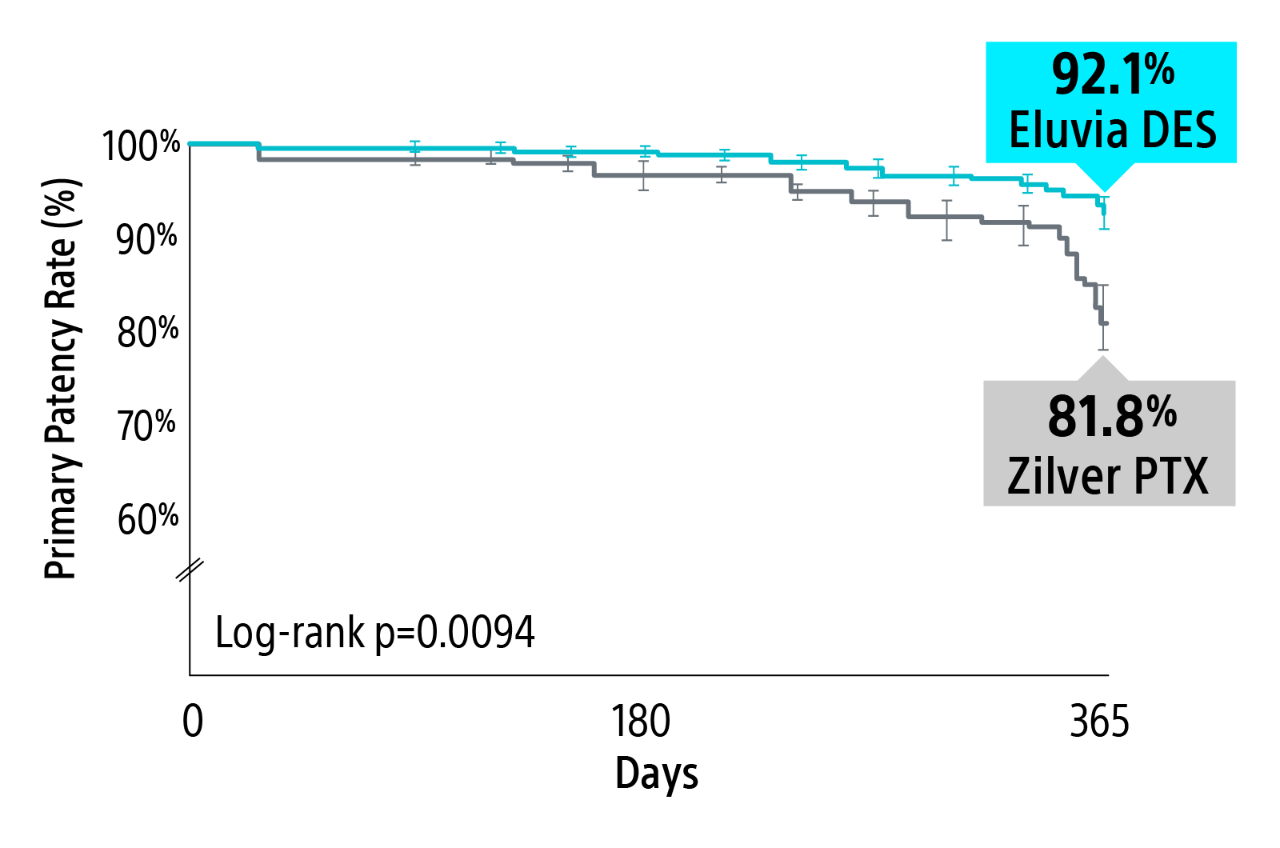
IMPERIAL RCT 1-year primary patency results
Eluvia Drug-eluting stent demonstrated superiority over Zilver PTX with a statistically significant primary patency through 1-Year.1
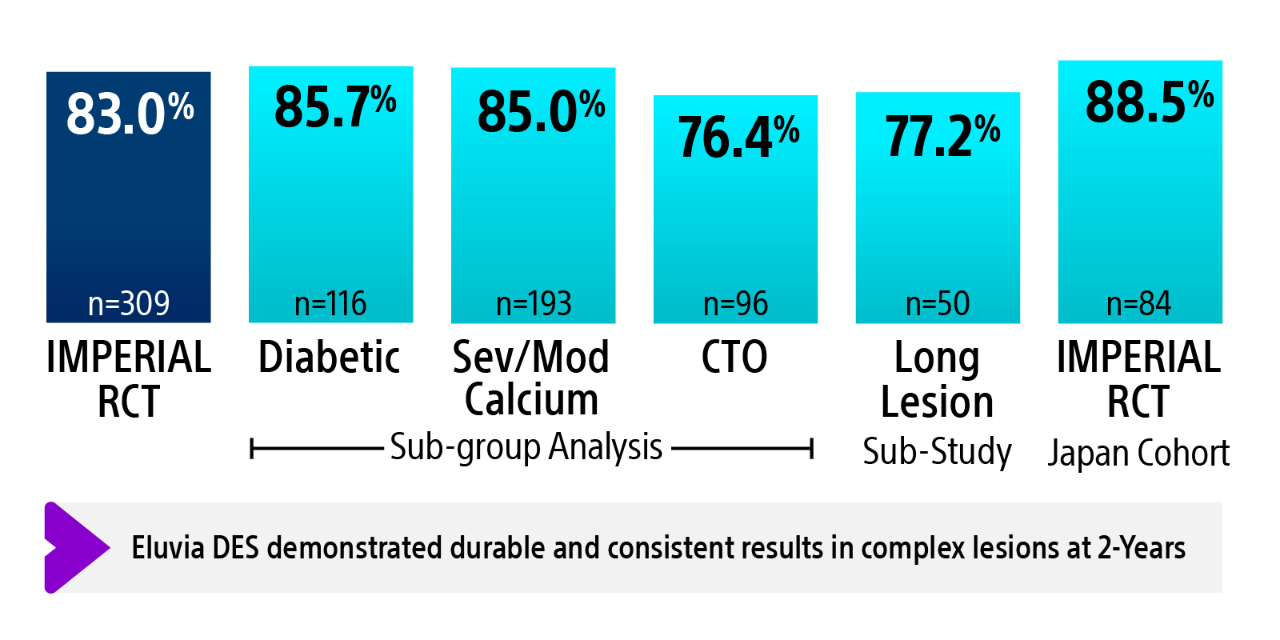
IMPERIAL RCT 2-year primary patency results2-6
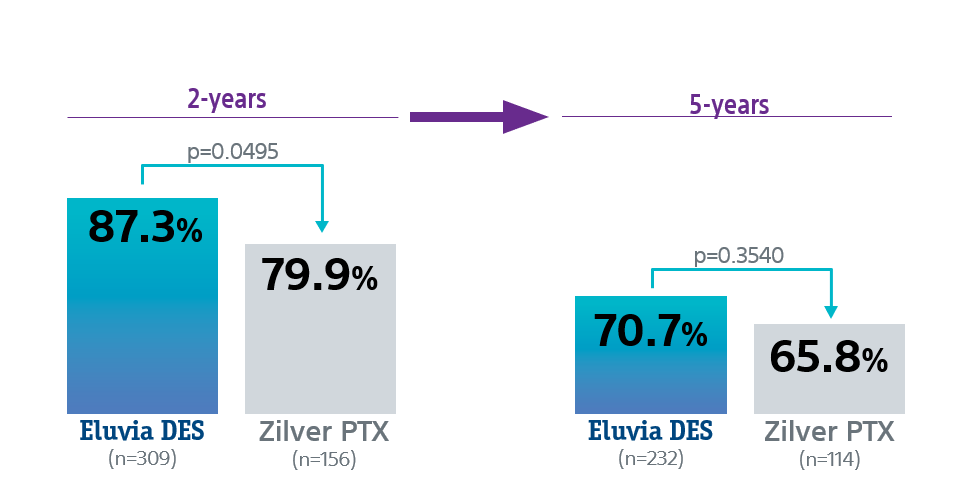
Freedom from CDTLR rates
Eluvia DES showed lower revascularization rates than Zilver PTX through 5 years with statistical significance at 2-Years.
IMPERIAL randomised controlled trial details
- 2-year primary endpoints
- Mortality rates
- Baseline characteristics
| 2-Year Primary Endpoints | Eluvia DES (n=309) | Zilver PTX (n=156) | p-value |
| Primary Patency | 83.0% | 77.1% | 0.1008 |
| Major Adverse Events | 14.2% | 20.1% | 0.1236 |
















