EMINENT randomized controlled trial results
EMINENT1 is the largest Randomized Controlled Trial (2:1) comparing Eluvia™ Drug-Eluting Vascular Stent System to self-expanding bare metal stents (BMS) for SFA/PPA; EU multi-center; superiority trial; core lab adjudicated.
EMINENT RCT 1-year primary patency results
Eluvia demonstrated superiority over BMS2 with a statistically significant primary patency of 85.4% versus 76.3% through 1-Year.
1-Year Kaplan-Meier primary patency estimate
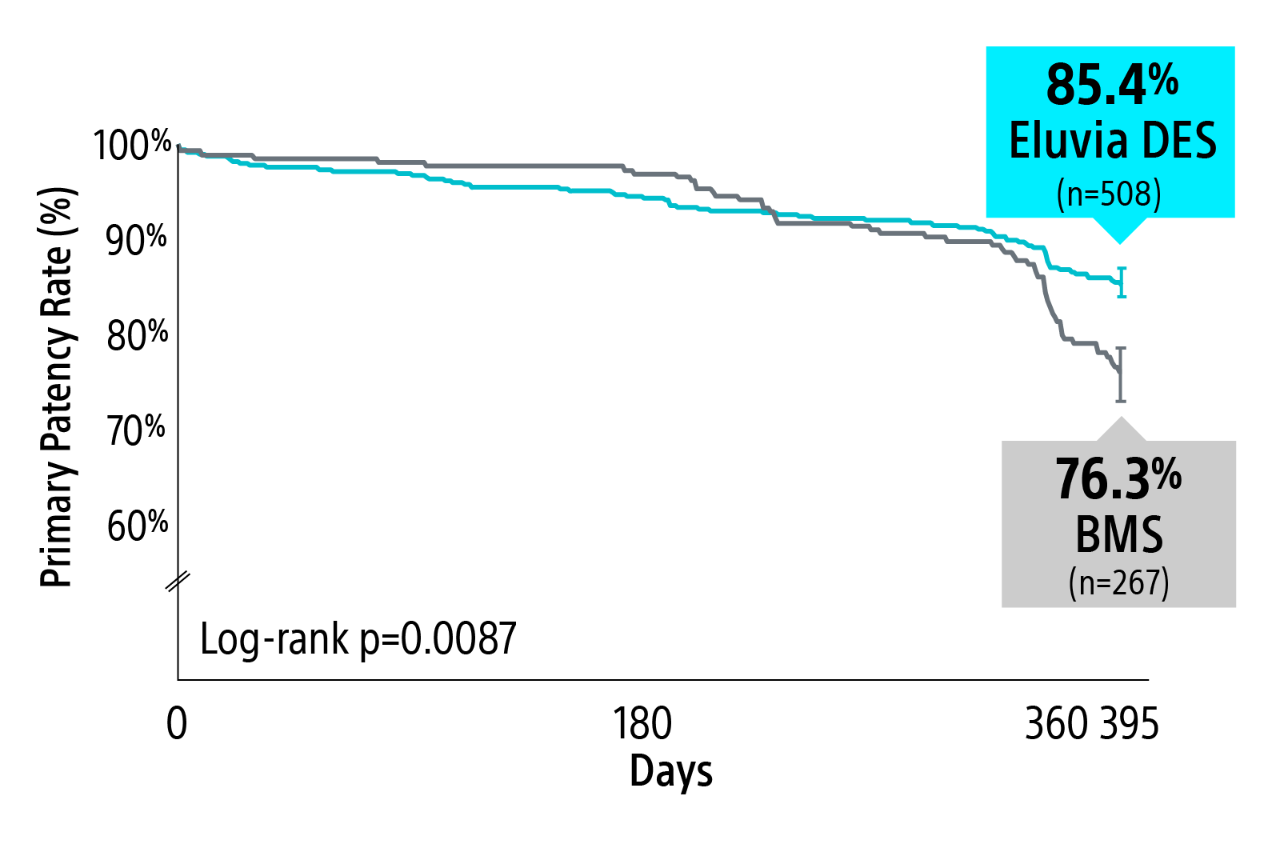
Kaplan-Meier Estimate: Primary patency defined as core-lab assessed duplex ultrasound peak systolic velocity ratio (PSVR) ≤ 2.4 at 1-year in the absence of clinically-driven TLR or bypass of the target lesion.
Log-rank p-value compares the entire K-M curves from time point zero to day 395 (full 1-year follow-up window).
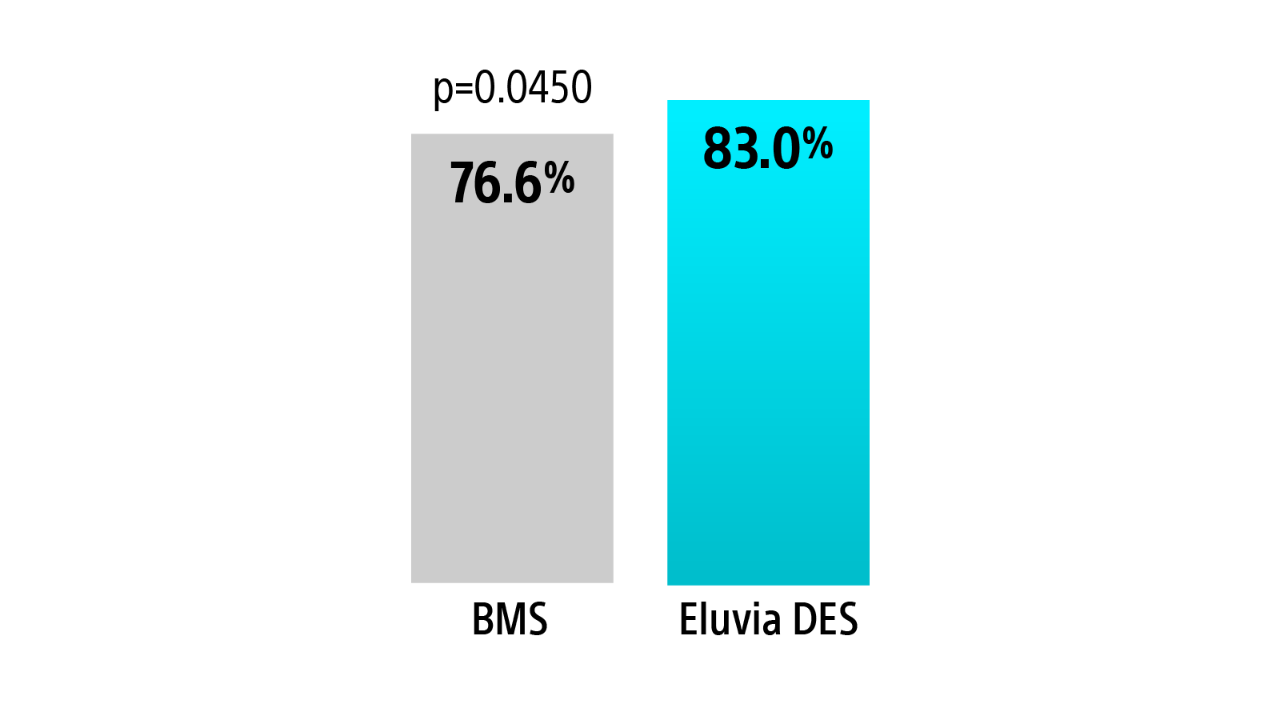
EMINENT RCT 1-year primary sustained clinical improvement
Eluvia demonstrated a statistically significant greater rate of sustained clinical improvement without reintervention over BMS through 1-Year.3
EMINENT randomized controlled trial details
- 1-year safety results
- Baseline characteristics
| Eluvia DES (n=492) | BMS (n=273) | p-value | |
| All Death, Major Amputation, TLR | 11.8% (56/474) | 11.8% (31/263) | 0.9912 |
| All-Cause Death at 1-Year | 2.7% (13/474) | 1.1% (3/263) | 0.1528 |
| Target Limb Major Amputation | 0.2% (1/474) | 0.0% (0/263) | 1.0000 |
| Clinically-Driven Target Lesion Revascularization | 8.4% (40/474) | 10.6% (28/263) | 0.3212 |