Boston Scientific accounts are for healthcare professionals only.



VersaCross™ Steerable Access Solution
Configure or select a product to continue to order
- Overview
- Clinical data
- Technical specifications
- Ordering information
- Training
- Resources
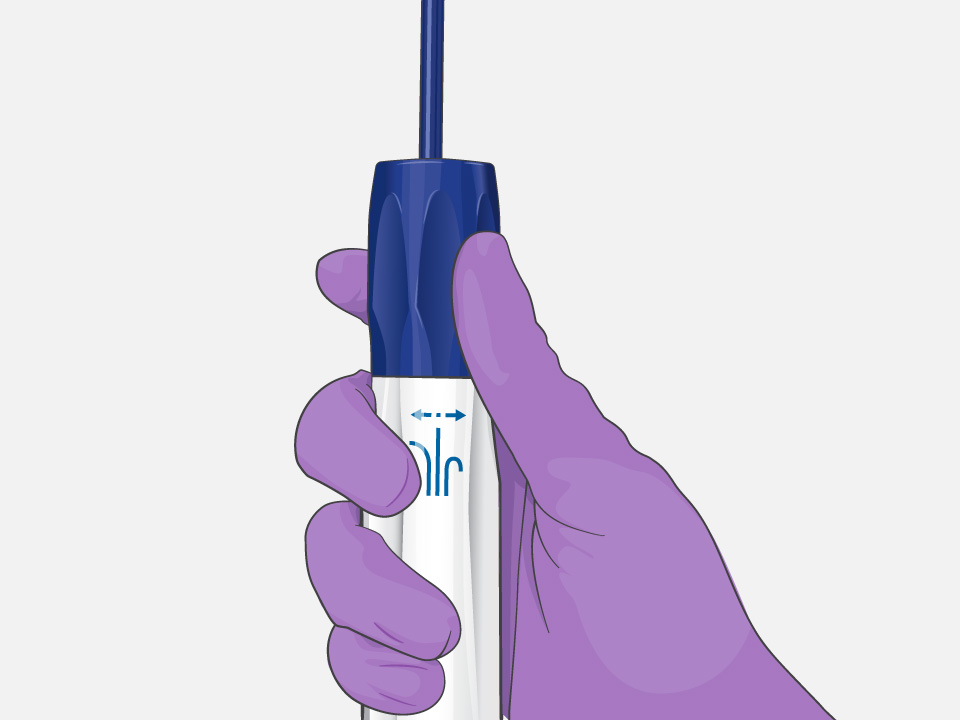
Deliver consistent contact force*†
Retain your curve and support consistent contact force
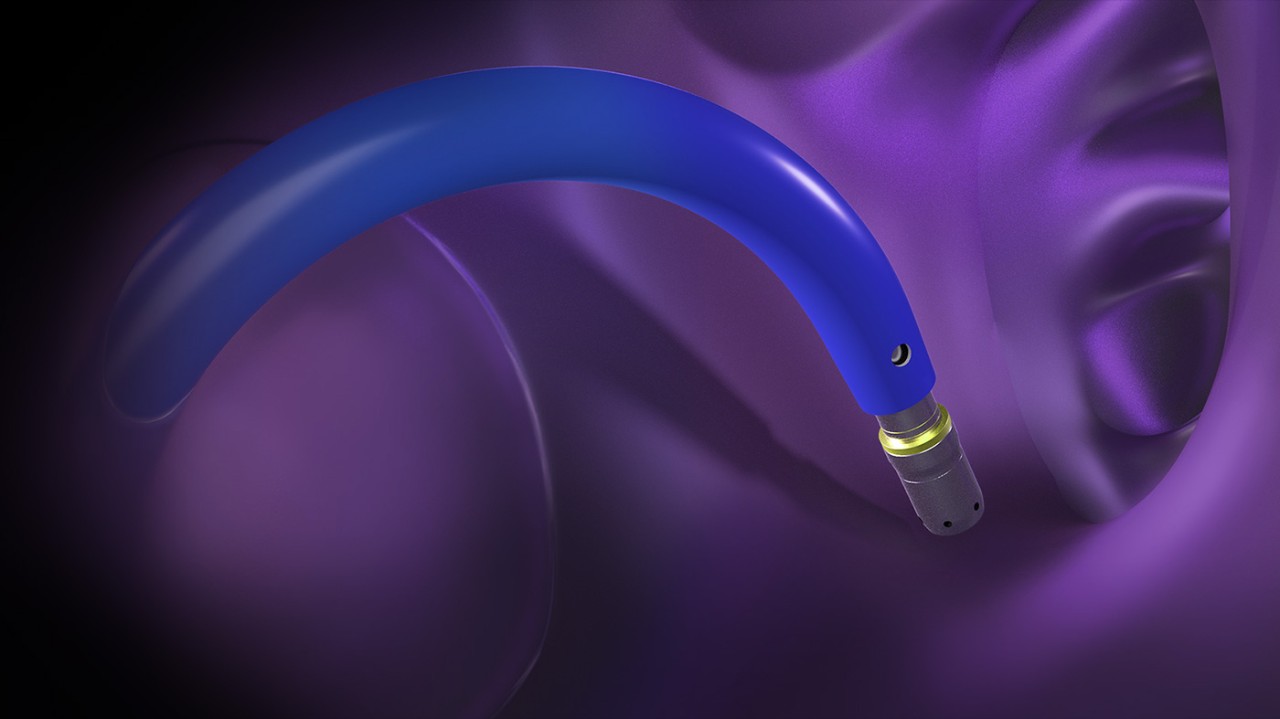
How it works
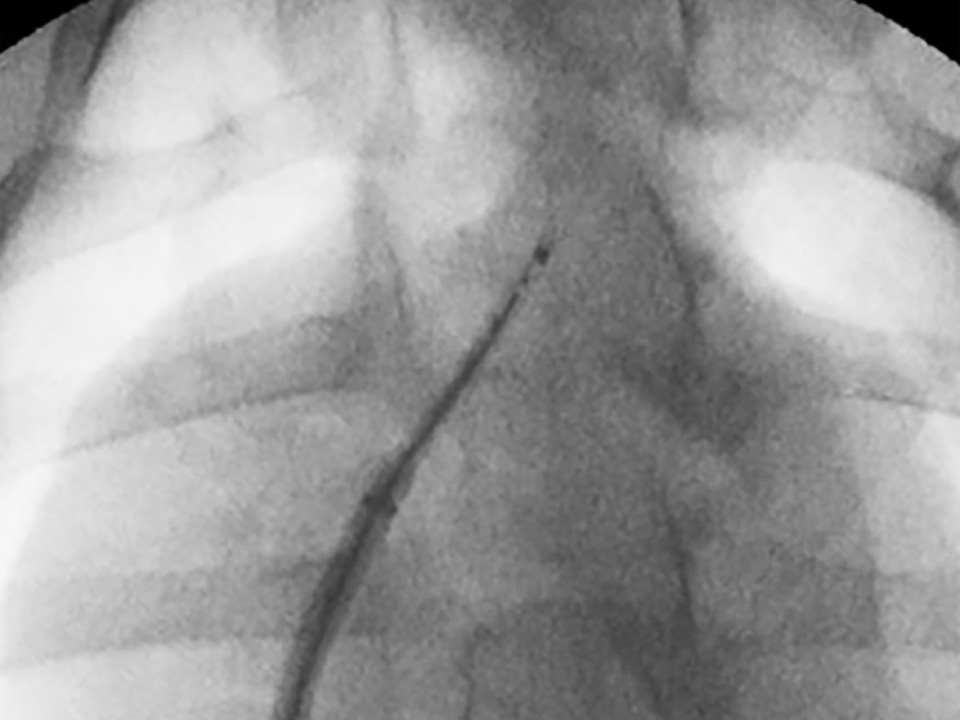
Clinical footage of the VersaCross Steerable Access Solution, an all-in-one device for transseptal access with high-precision steering
Results from case studies are not necessarily predictive of results in other cases. Results in other cases may vary.
Why choose the VersaCross Steerable Access Solution
Precise steering
Three solutions, one device
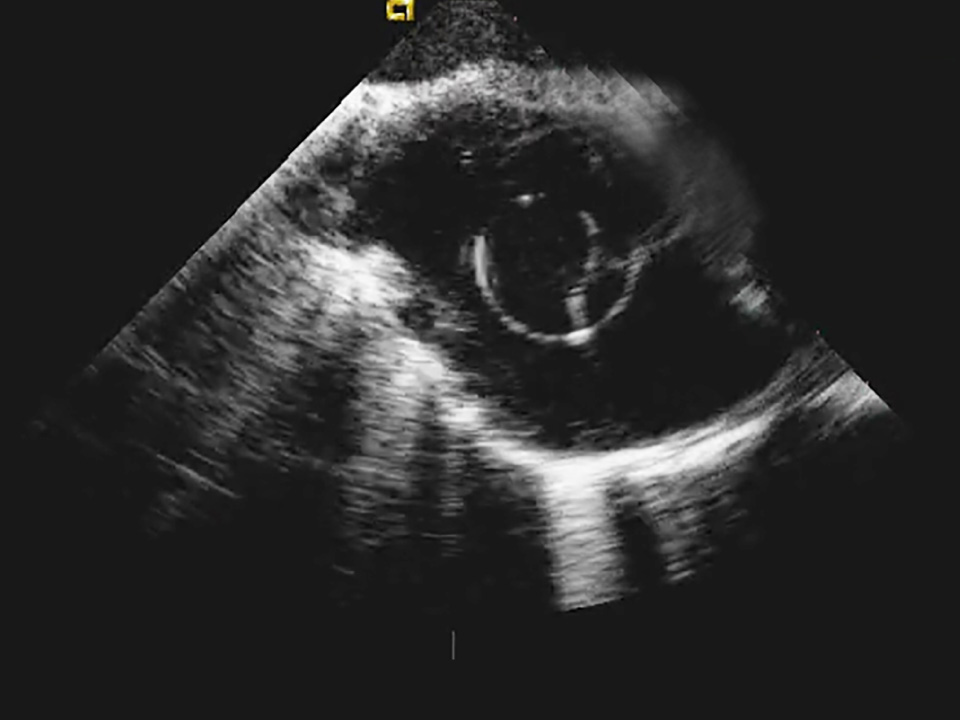
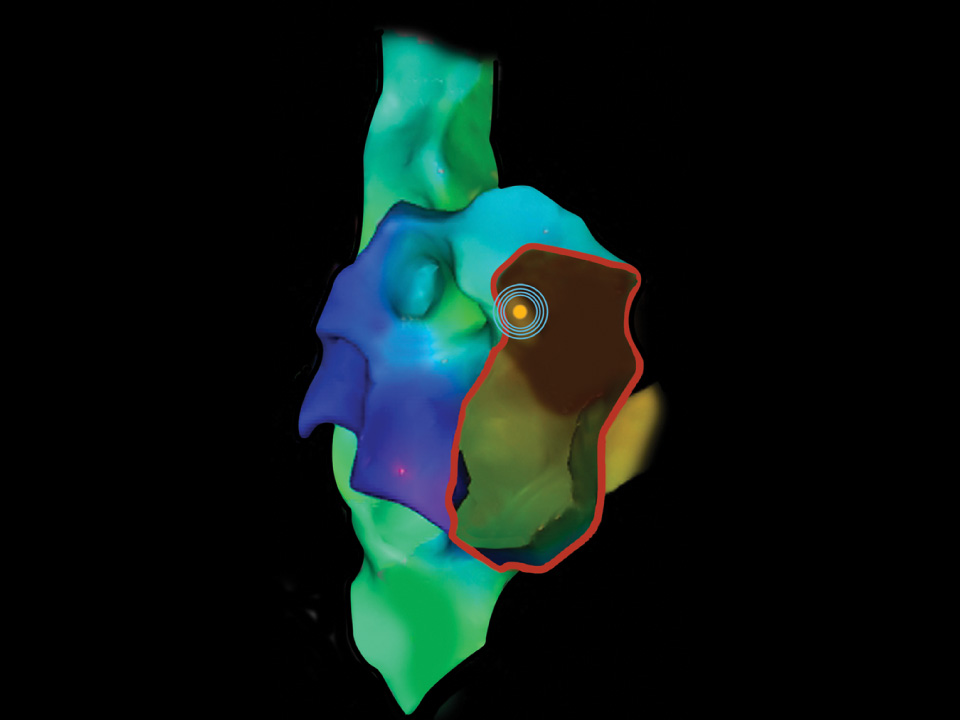
Know where you are at all times
OMNIviz™ technology enhances visualization on fluoroscopy, ultrasound, and electrical anatomical mapping
What’s included
The VersaCross Steerable Access Solution includes:
- VersaCross RF Wire (j-tip or pigtail)
- Single-use RFP-100A Connector Cable
- VersaCross Steerable Sheath (8.5F)
- Dilator with TRUform™ shapeable technology
- 0.035” guidewire
You can also purchase the VersaCross Steerable Sheath on its own.
Required product

Designed for controlled tissue puncture using radiofrequency energy
* Assessed by full bidirectional articulation to maximal extension up to 10 times to achieve mechanical fatigue (p=0.007). Comparison done using five SureFlex™ Steerable Guiding Sheaths and three competitor sheaths, with needle and dilator inside sheath.
† Based on bench testing conducted using SureFlex Steerable Guiding Sheath, which has identical steering mechanisms to VersaCross Steerable Sheath. Bench testing or pre-clinical study results may not necessarily be indicative of clinical performance. The testing was performed by or on behalf of Boston Scientific. Data on file.
‡ Assessed by handle rotation to a full 360° to test axial torque transfer and the maximum rotational capabilities of each sheath. Comparison done using five SureFlex™ sheaths and three competitor sheaths.
§ Assessed by a single pull to failure for the first test (27 SureFlex and five competitor sheaths), and repeated unidirectional articulation until failure or up to 300 cycles for the second test (five SureFlex and six competitor sheaths). Comparison done with needle and dilator inside sheath.
‖ Baylis Medical Company Radiofrequency Puncture Generator RFP-100A. Baylis Medical Company is a wholly owned subsidiary of Boston Scientific Corporation.
All trademarks are the property of their respective owners.