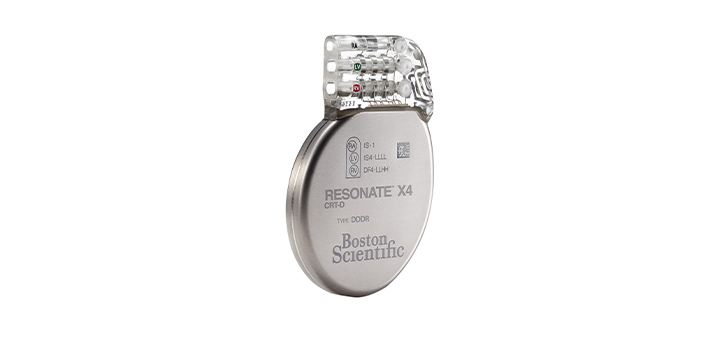
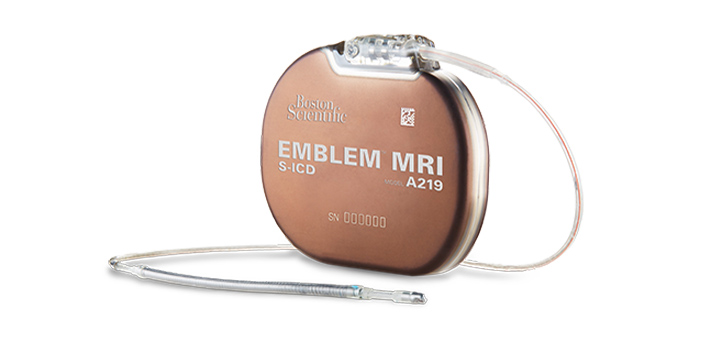
Our focus
Groundbreaking technologies that monitor, support diagnosis, and treat irregular heart rhythms, heart failure and sudden cardiac arrest.
Kevin’s story
We are collaborating with healthcare professionals to help improve outcomes, enhance patients' lives, and reduce costs with the most advanced and least invasive cardiac rhythm management monitoring and therapeutic technologies available.