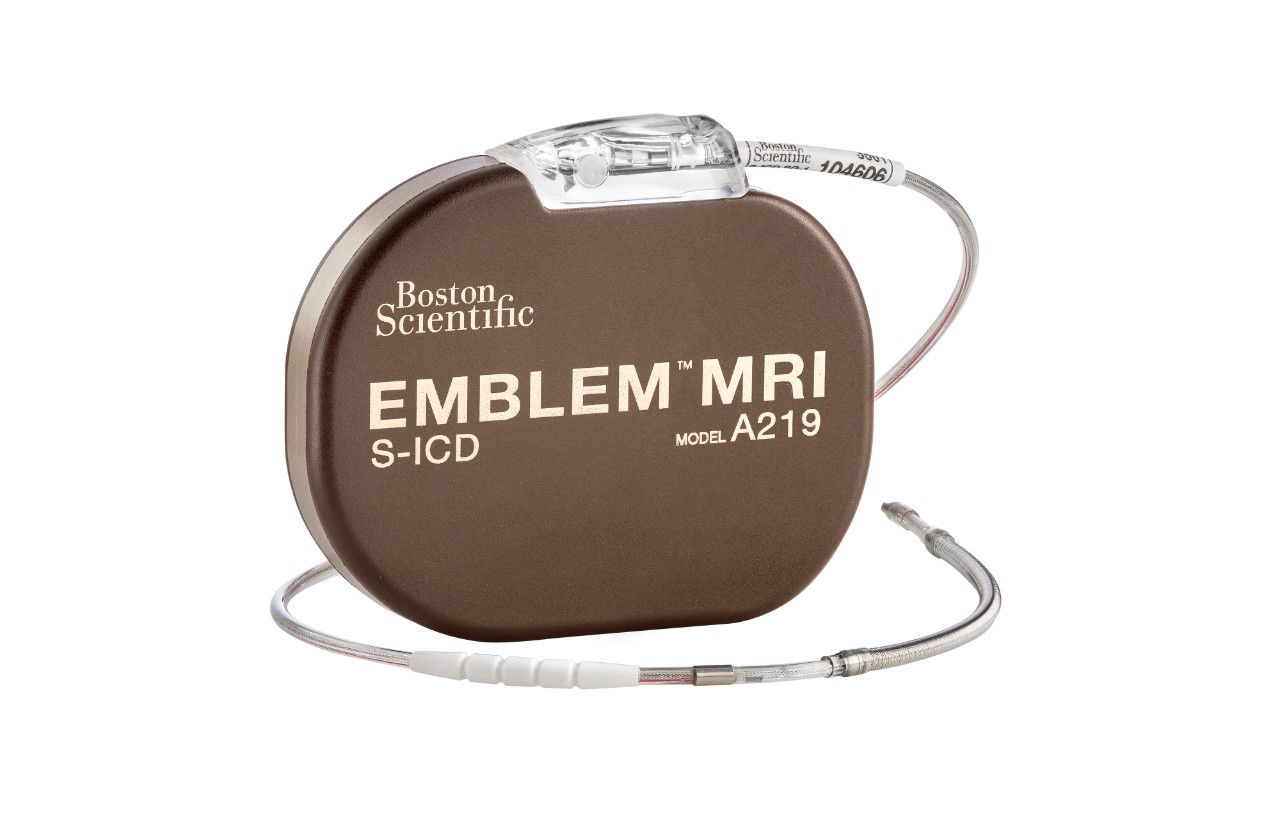
MRI Compatibility
The EMBLEM™ MRI and EMBLEM™ S-ICDs are FDA-approved for use with any S-ICD electrode as an ImageReady MR-Conditional System.
- Full body scans without time restrictions in 1.5T MRI environments (SAR <=2.0W/Kg whole body averaged, SAR<=3.2 head @/kg)
- MRI Protection Mode available in programmer with automatic time-out feature
The EMBLEM MRI S-ICD system provides effective defibrillation without transvenous leads.
- Eliminates complications associated with transvenous leads
- Reduces potential for systemic infection or endocarditis
- Eliminates potential for vascular or cardiac injuries
- Preserves vasculature for potential future interventions
Model Information
| Product Name | Model Number |
|---|---|
| EMBLEM MRI S-ICD | A219 |
| EMBLEM S-ICD | A209 |
| S-ICD Electrode | 3010, 3400, 3401, 3501 |